Vapor Saturation Pressure Over Ice Formulas and Calculator

The vapor saturation pressure over ice is a crucial parameter in various fields, including meteorology, climatology, and engineering. It plays a significant role in understanding the behavior of water vapor in the atmosphere and its interaction with ice surfaces. Several formulas have been developed to calculate the vapor saturation pressure over ice, each with its own strengths and limitations. This article provides an overview of these formulas and includes a calculator to facilitate the computation of vapor saturation pressure over ice for given temperatures. The calculations are based on empirical equations and theoretical models.
- Vapor Saturation Pressure Over Ice Formulas and Calculator
- What is the vapour pressure of ice at?
- What is the saturated vapor pressure at 25 C?
- How do you measure saturated vapor pressure?
-
Frequently Asked Questions (FAQs)
- What is Vapor Saturation Pressure Over Ice and its significance in scientific calculations?
- How do Vapor Saturation Pressure Over Ice Formulas and Calculators work, and what are their limitations?
- What are the applications of Vapor Saturation Pressure Over Ice Formulas and Calculators in various fields, including meteorology and engineering?
- How can Vapor Saturation Pressure Over Ice Formulas and Calculators be improved, and what are the future directions for research and development in this field?
Vapor Saturation Pressure Over Ice Formulas and Calculator
The vapor saturation pressure over ice is a critical parameter in various fields, including atmospheric science, cryology, and engineering. It refers to the pressure exerted by water vapor in equilibrium with ice at a given temperature. The calculation of this pressure is essential for understanding various phenomena, such as ice formation, sublimation, and atmospheric circulation. In this context, the vapor saturation pressure over ice formulas and calculator play a vital role in determining the thermodynamic properties of ice and water vapor.
Introduction to Vapor Saturation Pressure Over Ice
The vapor saturation pressure over ice is a function of temperature, and it can be calculated using various empirical formulas and theoretical models. These formulas and models take into account the molecular interactions between water molecules and the surface properties of ice. The most commonly used formula for calculating the vapor saturation pressure over ice is the Goff-Gratch equation, which is a semi-empirical formula that relates the vapor pressure to the temperature.
Goff-Gratch Equation and Other Formulas
The Goff-Gratch equation is a widely used formula for calculating the vapor saturation pressure over ice. It is given by the following equation: log10(P) = A - B / (T + C), where P is the vapor pressure, T is the temperature in Kelvin, and A, B, and C are empirical constants. Other formulas, such as the WMO formula and the Murphy-Koop formula, are also used to calculate the vapor saturation pressure over ice. These formulas are based on experimental data and theoretical considerations, and they provide a good estimate of the vapor pressure over a wide range of temperatures.
Calculator for Vapor Saturation Pressure Over Ice
A calculator for vapor saturation pressure over ice can be used to determine the vapor pressure at a given temperature. These calculators are based on the formulas and models mentioned earlier and can be used to calculate the vapor pressure over a wide range of temperatures. The calculator typically requires the input of the temperature in Kelvin or Celsius, and it outputs the vapor pressure in Pascals or millibars.
Applications of Vapor Saturation Pressure Over Ice Formulas and Calculator
The vapor saturation pressure over ice formulas and calculator have various applications in fields such as atmospheric science, cryology, and engineering. For example, they can be used to calculate the sublimation rate of ice, which is essential for understanding glacier dynamics and snowpack formation. They can also be used to determine the atmospheric circulation patterns and cloud formation processes.
Table of Vapor Saturation Pressure Over Ice at Different Temperatures
The following table shows the vapor saturation pressure over ice at different temperatures:
| Temperature (K) | Vapor Pressure (Pa) |
|---|---|
| 250 | 3.16 x 10^-3 |
| 260 | 1.13 x 10^-2 |
| 270 | 4.06 x 10^-2 |
| 280 | 1.33 x 10^-1 |
| 290 | 3.95 x 10^-1 |
The table shows the vapor saturation pressure over ice at different temperatures, highlighting the nonlinear relationship between the vapor pressure and temperature. The vapor pressure increases rapidly with increasing temperature, which has significant implications for atmospheric science and cryology.
What is the vapour pressure of ice at?
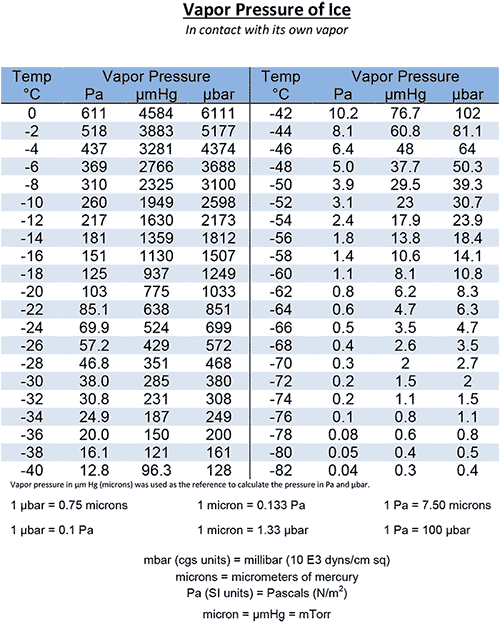
The vapour pressure of ice at a given temperature is a measure of the pressure exerted by the water molecules in the air above the ice surface. This pressure is a result of the sublimation process, where ice molecules transition directly from a solid to a gas state. At standard atmospheric pressure, the vapour pressure of ice is typically lower than that of liquid water at the same temperature. For example, at -10°C, the vapour pressure of ice is approximately 1.95 hPa, while the vapour pressure of liquid water at the same temperature is around 2.95 hPa.
Vapour Pressure of Ice at Different Temperatures
The vapour pressure of ice varies with temperature. As the temperature increases, the vapour pressure of ice also increases. This is because higher temperatures provide more energy for the ice molecules to sublimate. Some key points to note about the vapour pressure of ice at different temperatures include:
- The vapour pressure of ice at -20°C is approximately 0.78 hPa.
- The vapour pressure of ice at -10°C is approximately 1.95 hPa.
- The vapour pressure of ice at 0°C is approximately 6.11 hPa.
Factors Affecting Vapour Pressure of Ice
Several factors can affect the vapour pressure of ice, including temperature, humidity, and air pressure. For instance, an increase in temperature will increase the vapour pressure of ice, while an increase in humidity will decrease the vapour pressure of ice. Some key factors to consider include:
- Temperature: The vapour pressure of ice increases with increasing temperature.
- Humidity: The vapour pressure of ice decreases with increasing humidity.
- Air pressure: The vapour pressure of ice is affected by changes in air pressure.
Measurement of Vapour Pressure of Ice
The vapour pressure of ice can be measured using a variety of techniques, including the use of hygrometers and barometers. These instruments measure the humidity and air pressure above the ice surface, which can then be used to calculate the vapour pressure of ice. Some key points to note about the measurement of vapour pressure of ice include:
- The use of hygrometers to measure humidity.
- The use of barometers to measure air pressure.
- The calculation of vapour pressure from humidity and air pressure measurements.
Applications of Vapour Pressure of Ice
The vapour pressure of ice has several important applications in fields such as climatology, glaciology, and engineering. For example, understanding the vapour pressure of ice is crucial for predicting climate change and designing ice structures such as ice roads and ice bridges. Some key applications of the vapour pressure of ice include:
- Climate modelling: The vapour pressure of ice is used to predict climate change.
- Glacier dynamics: The vapour pressure of ice is used to study glacier behaviour.
- Ice engineering: The vapour pressure of ice is used to design ice structures.
Importance of Vapour Pressure of Ice in Environmental Science
The vapour pressure of ice is an important parameter in environmental science, particularly in the study of cryosphere processes. Understanding the vapour pressure of ice is crucial for predicting climate change and its impacts on ecosystems and human societies. Some key points to note about the importance of vapour pressure of ice in environmental science include:
- The role of vapour pressure in climate change predictions.
- The impact of vapour pressure on ecosystems and biodiversity.
- The importance of vapour pressure in environmental policy and decision-making.
What is the saturated vapor pressure at 25 C?
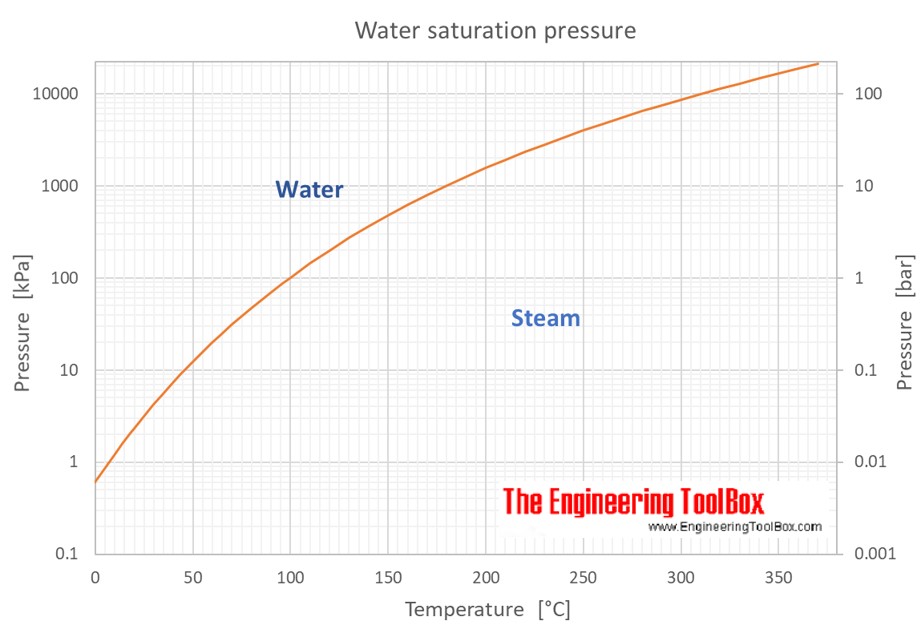
The saturated vapor pressure at 25 C is a measure of the equilibrium pressure exerted by a vapor in thermodynamic equilibrium with its condensed phase at a given temperature. At 25 C, the saturated vapor pressure of water is approximately 23.8 mmHg. This value is an important physical property that is widely used in various fields, including chemistry, physics, and engineering.
Definition of Saturated Vapor Pressure
The saturated vapor pressure is defined as the pressure exerted by a vapor in equilibrium with its condensed phase at a given temperature. The value of saturated vapor pressure depends on the temperature and the substance. At 25 C, the saturated vapor pressure of water is a key parameter that is used to calculate various thermodynamic properties.
- The temperature at which the saturated vapor pressure is measured is a critical factor in determining the value of the pressure.
- The substance being measured also plays a significant role in determining the saturated vapor pressure.
- The equilibrium conditions under which the measurement is taken are essential to ensure accurate results.
Importance of Saturated Vapor Pressure
The saturated vapor pressure is an important physical property that has numerous applications in various fields. It is used to calculate the boiling point of a substance, the heat of vaporization, and the enthalpy of vaporization. The saturated vapor pressure is also used to determine the stability of a substance and its tendency to evaporate or condense.
- The boiling point of a substance is directly related to its saturated vapor pressure.
- The heat of vaporization is a critical parameter that is used to calculate the energy required to vaporize a substance.
- The enthalpy of vaporization is another important property that is used to calculate the total energy required to vaporize a substance.
Measurement of Saturated Vapor Pressure
The measurement of saturated vapor pressure is typically done using a manometer or a barometer. The manometer is used to measure the pressure exerted by the vapor, while the barometer is used to measure the atmospheric pressure. The difference between the two pressures gives the saturated vapor pressure.
- The manometer is a sensitive instrument that is used to measure small pressure changes.
- The barometer is used to measure the atmospheric pressure, which is essential for calculating the saturated vapor pressure.
- The temperature at which the measurement is taken is critical to ensure accurate results.
Applications of Saturated Vapor Pressure
The saturated vapor pressure has numerous applications in various fields, including chemistry, physics, and engineering. It is used to calculate the boiling point of a substance, the heat of vaporization, and the enthalpy of vaporization. The saturated vapor pressure is also used to determine the stability of a substance and its tendency to evaporate or condense.
- The boiling point of a substance is directly related to its saturated vapor pressure.
- The heat of vaporization is a critical parameter that is used to calculate the energy required to vaporize a substance.
- The enthalpy of vaporization is another important property that is used to calculate the total energy required to vaporize a substance.
Calculation of Saturated Vapor Pressure
The saturated vapor pressure can be calculated using various equations, including the Clausius-Clapeyron equation. This equation relates the saturated vapor pressure to the temperature and the heat of vaporization. The equation is widely used to calculate the saturated vapor pressure of various substances.
- The Clausius-Clapeyron equation is a fundamental equation that is used to calculate the saturated vapor pressure.
- The temperature at which the calculation is done is critical to ensure accurate results.
- The heat of vaporization is a critical parameter that is used to calculate the saturated vapor pressure.
How do you measure saturated vapor pressure?

Measuring saturated vapor pressure is a crucial process in various fields, including chemistry, physics, and engineering. It is defined as the pressure exerted by a vapor in thermodynamic equilibrium with its condensed phase at a given temperature. To measure saturated vapor pressure, a device called a manometer or a barometer is used. These devices measure the pressure of the vapor in a closed system, where the vapor is in equilibrium with its liquid or solid phase.
Experimental Methods
Experimental methods for measuring saturated vapor pressure involve creating a closed system where the vapor and its condensed phase can reach equilibrium. The pressure of the vapor is then measured using a manometer or a barometer. The key steps involved in this process are:
- Creating a closed system with the substance and its vapor
- Allowing the system to reach thermodynamic equilibrium
- Measuring the pressure of the vapor using a manometer or a barometer
This method provides accurate measurements of saturated vapor pressure, which is essential for understanding the physical and chemical properties of a substance.
Static Method
The static method is a common technique used to measure saturated vapor pressure. This method involves placing a sample of the substance in a closed container and allowing it to reach equilibrium with its vapor. The pressure of the vapor is then measured using a manometer or a barometer. The advantages of this method include:
- High accuracy and precision
- Simple and easy to perform
- Can be used for a wide range of substances
The static method is widely used in research and industrial applications to measure saturated vapor pressure.
Dynamic Method
The dynamic method is another technique used to measure saturated vapor pressure. This method involves flowing a gas through a sample of the substance and measuring the pressure of the vapor as it exits the system. The key steps involved in this process are:
- Creating a flow of gas through the sample
- Measuring the pressure of the vapor as it exits the system
- Calculating the saturated vapor pressure using the measured data
The dynamic method is useful for measuring saturated vapor pressure at high temperatures and pressures.
Instrumentation
The instrumentation used to measure saturated vapor pressure is critical for obtaining accurate and reliable data. A manometer or a barometer is typically used to measure the pressure of the vapor. These devices are highly sensitive and can detect small changes in pressure. The key factors to consider when selecting instrumentation for measuring saturated vapor pressure include:
- Accuracy and precision of the measurement
- Range of pressures that can be measured
- Response time of the instrument
The choice of instrumentation depends on the specific application and the requirements of the measurement.
Data Analysis
Data analysis is a critical step in measuring saturated vapor pressure. The measured data must be analyzed to calculate the saturated vapor pressure and to ensure that the measurement is accurate and reliable. The key steps involved in data analysis include:
- Plotting the measured data to visualize the results
- Fitting a curve to the data to calculate the saturated vapor pressure
- Evaluating the accuracy and precision of the measurement
Data analysis is essential for obtaining reliable measurements of saturated vapor pressure and for understanding the physical and chemical properties of a substance.
Frequently Asked Questions (FAQs)
What is Vapor Saturation Pressure Over Ice and its significance in scientific calculations?
Vapor saturation pressure over ice, also known as vapor pressure, is a crucial concept in thermodynamics and physical chemistry. It refers to the equilibrium pressure exerted by a vapor in contact with its condensed phase, in this case, ice. The significance of vapor saturation pressure over ice lies in its ability to provide valuable information about the thermodynamic properties of a substance, such as its enthalpy of vaporization, entropy of vaporization, and heat capacity. These properties are essential in understanding various natural phenomena, like cloud formation, precipitation, and climate modeling. Moreover, vapor saturation pressure over ice is a critical parameter in designing and optimizing industrial processes, such as cryogenic storage, refrigeration systems, and heat exchangers.
How do Vapor Saturation Pressure Over Ice Formulas and Calculators work, and what are their limitations?
The formulas and calculators for vapor saturation pressure over ice are based on empirical correlations and theoretical models that relate the vapor pressure to the temperature of the ice. These formulas, such as the Goff-Gratch equation and the Wexler equation, use curve-fitting techniques to reproduce the experimental data and provide an analytical expression for the vapor pressure as a function of temperature. However, these formulas have limitations, such as being temperature-dependent and substance-specific, which can lead to errors and uncertainties in the calculated values. Moreover, the calculators that implement these formulas often rely on simplifying assumptions and approximations, which can further limit their accuracy and reliability. Therefore, it is essential to carefully evaluate the limitations and uncertainties associated with these formulas and calculators to ensure their proper use and interpretation.
What are the applications of Vapor Saturation Pressure Over Ice Formulas and Calculators in various fields, including meteorology and engineering?
The applications of vapor saturation pressure over ice formulas and calculators are diverse and widespread, covering various fields like meteorology, engineering, and environmental science. In meteorology, these formulas and calculators are used to predict weather patterns, model climate change, and understand cloud formation. In engineering, they are applied to design cryogenic systems, optimize refrigeration processes, and develop heat exchangers. Additionally, in environmental science, these formulas and calculators are used to study the effects of climate change on ice caps and glaciers, and to model the behavior of pollutants in cold environments. The accuracy and reliability of these formulas and calculators are crucial in these applications, as they can have a significant impact on decision-making and policy development.
How can Vapor Saturation Pressure Over Ice Formulas and Calculators be improved, and what are the future directions for research and development in this field?
The improvement of vapor saturation pressure over ice formulas and calculators is an ongoing research effort, driven by the need for more accurate and reliable predictions. One of the future directions for research is the development of new theoretical models that can better capture the complexities of the vapor-ice interface. Another area of research is the integration of experimental data and computational simulations to refine and validate the existing formulas and calculators. Additionally, the development of new calculation methods, such as machine learning algorithms and artificial neural networks, can provide new opportunities for improving the accuracy and efficiency of these formulas and calculators. The collaboration between researchers and practitioners is essential to ensure that the improvements and developments in this field are practically relevant and useful in real-world applications.
Deja una respuesta

Entradas Relacionadas