Heat Required to Increase Temperature of Reservoir Formula and Calculator
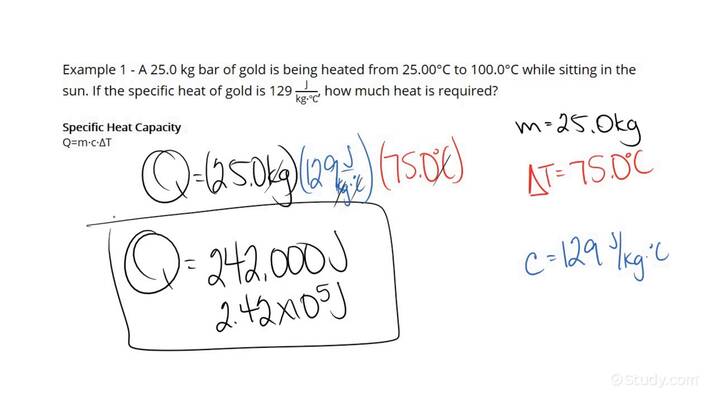
The amount of heat required to increase the temperature of a reservoir can be calculated using a specific formula. This formula takes into account the mass of the water, the specific heat capacity of water, and the desired temperature change. The calculation is crucial in various engineering and scientific applications, such as heating systems, thermal energy storage, and water treatment processes. Understanding the formula and using a calculator can help professionals and researchers determine the exact amount of heat needed to achieve a specific temperature increase in a reservoir. This facilitates efficient and accurate calculations.
- Heat Required to Increase Temperature of Reservoir Formula and Calculator
- How to calculate heat required to raise the temperature of water?
- What is the formula for the heat required to raise the temperature?
- What is the formula for raising temperature?
-
Frequently Asked Questions (FAQs)
- What is the formula for calculating the heat required to increase the temperature of a reservoir?
- How does the specific heat capacity of water affect the heat required to increase the temperature of a reservoir?
- What are the factors that affect the heat required to increase the temperature of a reservoir?
- How can the heat required to increase the temperature of a reservoir be calculated using a calculator?
Heat Required to Increase Temperature of Reservoir Formula and Calculator
The heat required to increase the temperature of a reservoir can be calculated using the formula Q = mcΔT, where Q is the amount of heat energy required, m is the mass of the reservoir, c is the specific heat capacity of the reservoir material, and ΔT is the change in temperature. This formula is a fundamental concept in thermodynamics and is widely used in various fields, including engineering, physics, and chemistry. The specific heat capacity of a material is a measure of its ability to absorb and release heat energy, and it is an important factor in determining the amount of heat required to increase the temperature of a reservoir.
Understanding the Formula and Calculator
The formula Q = mcΔT is a simple yet powerful tool for calculating the heat required to increase the temperature of a reservoir. The calculator is an online tool that can be used to perform the calculation quickly and easily. To use the calculator, the user simply needs to enter the mass of the reservoir, the specific heat capacity of the reservoir material, and the change in temperature. The calculator then performs the calculation and displays the result.
Importance of Specific Heat Capacity
The specific heat capacity of a material is a critical factor in determining the amount of heat required to increase the temperature of a reservoir. Materials with high specific heat capacities, such as water, require more heat energy to change their temperature than materials with low specific heat capacities, such as air. The specific heat capacity of a material can be found in reference tables or by performing experiments.
Applications of the Formula and Calculator
The formula Q = mcΔT and the calculator have a wide range of applications in various fields, including engineering, physics, and chemistry. For example, they can be used to design heating systems, calculate the energy required to heat a building or a process, and determine the thermal energy required to raise the temperature of a material. They can also be used in research and development to study the thermal properties of materials and to develop new technologies.
Limitations and Assumptions
The formula Q = mcΔT and the calculator are based on several assumptions and limitations. For example, they assume that the reservoir is a closed system, meaning that no heat energy is transferred to or from the surroundings. They also assume that the specific heat capacity of the reservoir material is constant over the temperature range of interest. In reality, the specific heat capacity of a material can vary with temperature, and this can affect the accuracy of the calculation.
Example Calculation
To illustrate the use of the formula and calculator, consider an example calculation. Suppose we want to calculate the heat required to raise the temperature of a 1000 kg reservoir of water from 20°C to 30°C. The specific heat capacity of water is approximately 4186 J/kg°C. Using the formula Q = mcΔT, we can calculate the heat required as follows:
| Parameter! | Value |
|---|---|
| Mass (m) | 1000 kg |
| Specific heat capacity (c) | 4186 J/kg°C |
| Change in temperature (ΔT) | 10°C |
| Heat required (Q) | 41,860,000 J |
The result shows that approximately 41,860,000 J of heat energy is required to raise the temperature of the reservoir from 20°C to 30°C.
How to calculate heat required to raise the temperature of water?

To calculate the heat required to raise the temperature of water, we need to use the specific heat capacity of water and the temperature difference. The specific heat capacity of water is approximately 4.184 joules per gram per degree Celsius. We can use the formula: Q = mcΔT, where Q is the heat required, m is the mass of water, c is the specific heat capacity, and ΔT is the temperature difference.
Understanding the Formula
The formula Q = mcΔT is used to calculate the heat required to raise the temperature of water. This formula takes into account the mass of water, the specific heat capacity, and the temperature difference. The mass of water is typically measured in grams or kilograms, while the temperature difference is measured in degrees Celsius. The specific heat capacity is a constant value that represents the amount of heat required to raise the temperature of a substance by one degree Celsius.
- The mass of water is an important factor in calculating the heat!
- The specific heat capacity of water is approximately 4.184 joules per gram per degree Celsius.
- The temperature difference is the difference between the initial and final temperatures of the water.
Calculating the Heat Required
To calculate the heat required to raise the temperature of water, we need to plug in the values into the formula Q = mcΔT. For example, if we want to raise the temperature of 1 kilogram of water from 20 degrees Celsius to 80 degrees Celsius, we would use the following values: m = 1 kilogram, c = 4.184 joules per gram per degree Celsius, and ΔT = 60 degrees Celsius. We would then calculate the heat required using the formula.
- Convert the mass of water from kilograms to grams.
- Calculate the temperature difference by subtracting the initial temperature from the final temperature.
- Plug in the values into the formula Q = mcΔT and calculate the heat required.
Factors Affecting the Calculation
There are several factors that can affect the calculation of the heat required to raise the temperature of water. These include the specific heat capacity of the water, the temperature difference, and the mass of water. The specific heat capacity of water can vary slightly depending on the temperature and pressure of the water. Additionally, the temperature difference can affect the calculation, as a larger temperature difference will require more heat.
- The specific heat capacity of water can vary slightly depending on the temperature and pressure.
- The temperature difference can affect the calculation, as a larger temperature difference will require more heat.
- The mass of water is an important factor in calculating the heat required.
Applications of the Calculation
The calculation of the heat required to raise the temperature of water has several applications in various fields. For example, it can be used to design heating systems for buildings, swimming pools, and industrial processes. It can also be used to calculate the energy required to heat water for domestic use, such as showering and cooking.
- The calculation can be used to design heating systems for buildings and industrial processes.
- It can be used to calculate the energy required to heat water for domestic use.
- The calculation is important for energy efficiency and cost savings.
Common Mistakes to Avoid
When calculating the heat required to raise the temperature of water, there are several common mistakes to avoid. These include using the wrong units for the mass and temperature, not accounting for heat loss, and not using the correct specific heat capacity value. It is also important to ensure that the temperature difference is accurate and that the mass of water is correctly measured.
- Using the wrong units for the mass and temperature can lead to inaccurate calculations.
- Not accounting for heat loss can result in an underestimation of the heat required.
- Using the correct specific heat capacity value is crucial for accurate calculations.
What is the formula for the heat required to raise the temperature?

The formula for the heat required to raise the temperature is given by Q = mcΔT, where Q is the amount of heat added to the system, m is the mass of the substance, c is the specific heat capacity of the substance, and ΔT is the change in temperature.
Understanding the Formula
The formula Q = mcΔT is a fundamental concept in thermodynamics, which describes the relationship between the amount of heat added to a system and the resulting change in temperature. The specific heat capacity of a substance is a measure of how much heat is required to raise the temperature of a unit mass of the substance by one degree. This value is typically expressed in units of joules per kilogram per degree Celsius (J/kg°C). Some examples of specific heat capacities include:
- Water: 4.184 J/g°C
- Air: 1.005 J/g°C
- Copper: 0.385 J/g°C
Factors Affecting the Formula
The formula Q = mcΔT is affected by several factors, including the mass of the substance, the specific heat capacity of the substance, and the change in temperature. The mass of the substance determines the amount of heat required to raise the temperature, while the specific heat capacity determines how much heat is required to raise the temperature of a unit mass of the substance. The change in temperature also affects the amount of heat required, as a larger change in temperature requires more heat.
Applying the Formula
The formula Q = mcΔT can be applied to a wide range of situations, including heating a substance, cooling a substance, and determining the energy required to change the temperature of a substance. For example, if you want to heat a cup of water from 20°C to 100°C, you can use the formula to calculate the amount of heat required. Some examples of applications include:
- Heating a building: calculating the amount of heat required to maintain a comfortable temperature
- Cooling a substance: calculating the amount of heat required to lower the temperature of a substance
- Energy production: calculating the amount of energy required to produce a certain amount of heat
Limitations of the Formula
The formula Q = mcΔT has several limitations, including the assumption that the specific heat capacity of the substance is constant over the range of temperatures being considered. In reality, the specific heat capacity of a substance can vary depending on the temperature and pressure. Additionally, the formula does not take into account other factors that can affect the heat transfer, such as convection and radiation. Some examples of limiting factors include:
- Phase changes: the heat required to change the phase of a substance, such as from liquid to gas
- Non-linear relationships: the relationship between heat and temperature is not always linear
- External factors: external factors, such as environmental conditions, can affect the heat transfer
Real-World Applications
The formula Q = mcΔT has many real-world applications, including heating and cooling systems, energy production, and materials science. For example, the formula can be used to calculate the amount of heat required to heat a building or to cool a substance. Some examples of real-world applications include:
- Power plants: calculating the amount of heat required to produce electricity
- Refrigeration systems: calculating the amount of heat required to cool a substance
- Materials processing: calculating the amount of heat required to change the properties of a material
What is the formula for raising temperature?

The formula for raising temperature is a complex one, as it depends on various factors such as the material being heated, the heat source, and the environmental conditions. However, a general formula can be derived from the first law of thermodynamics, which states that the change in internal energy of a system is equal to the heat added to the system minus the work done by the system. Mathematically, this can be represented as Q = mcΔT, where Q is the heat energy, m is the mass of the material, c is the specific heat capacity, and ΔT is the change in temperature.
Understanding the Variables
To raise the temperature of a substance, it is essential to understand the variables involved in the formula. The specific heat capacity of a material is a measure of the amount of heat energy required to raise the temperature of a unit mass of the material by one degree. The mass of the material is also a critical factor, as it determines the amount of heat energy required to achieve a given temperature change. The change in temperature is the difference between the initial and final temperatures of the material.
- The specific heat capacity of a material can be found in reference tables or measured experimentally.
- The mass of the material can be measured using a balance or other weighing device.
- The change in temperature can be measured using a thermometer or other temperature-sensing device.
Heat Transfer Mechanisms
There are several heat transfer mechanisms that can be used to raise the temperature of a substance, including conduction, convection, and radiation. Conduction occurs when heat energy is transferred between particles in physical contact, while convection occurs when heat energy is transferred through the movement of fluids. Radiation is the transfer of heat energy through electromagnetic waves.
- Conduction is the most common heat transfer mechanism in solids.
- Convection is the most common heat transfer mechanism in fluids.
- Radiation is the most common heat transfer mechanism in gases.
Temperature Measurement
Temperature measurement is a critical aspect of raising the temperature of a substance. There are several types of temperature sensors available, including thermocouples, thermistors, and infrared sensors. The choice of temperature sensor depends on the temperature range and the accuracy required.
- Thermocouples are the most common type of temperature sensor.
- Thermistors are more accurate than thermocouples but have a limited temperature range.
- Infrared sensors are non-contact temperature sensors that use infrared radiation to measure temperature.
Heating Methods
There are several heating methods that can be used to raise the temperature of a substance, including electrical resistance heating, induction heating, and microwave heating. Electrical resistance heating uses an electric current to heat a resistive material, while induction heating uses an electromagnetic field to heat a conductive material. Microwave heating uses microwave radiation to heat a dielectric material.
- Electrical resistance heating is the most common heating method.
- Induction heating is more efficient than electrical resistance heating but requires a conductive material.
- Microwave heating is faster than other heating methods but can be difficult to control.
Applications of Temperature Control
Temperature control is essential in many industries, including chemical processing, food processing, and pharmaceutical manufacturing. Temperature control is used to optimize chemical reactions, prevent spoilage, and ensure product quality.
- Chemical processing requires precise temperature control to optimize chemical reactions.
- Food processing requires temperature control to prevent spoilage and ensure food safety.
- Pharmaceutical manufacturing requires temperature control to ensure product quality and efficacy.
Frequently Asked Questions (FAQs)
What is the formula for calculating the heat required to increase the temperature of a reservoir?
The formula for calculating the heat required to increase the temperature of a reservoir is Q = mcΔT, where Q is the amount of heat energy required, m is the mass of the water in the reservoir, c is the specific heat capacity of water, and ΔT is the change in temperature. This formula is based on the principle that the amount of heat energy required to change the temperature of a substance is directly proportional to its mass and specific heat capacity, and directly proportional to the change in temperature. The specific heat capacity of water is approximately 4.184 Joules per gram per degree Celsius, which means that it takes approximately 4.184 Joules of heat energy to raise the temperature of 1 gram of water by 1 degree Celsius.
How does the specific heat capacity of water affect the heat required to increase the temperature of a reservoir?
The specific heat capacity of water plays a crucial role in determining the amount of heat energy required to increase the temperature of a reservoir. The specific heat capacity of a substance is a measure of the amount of heat energy required to change its temperature by a given amount. In the case of water, its high specific heat capacity means that it can absorb and release large amounts of heat energy without undergoing a significant change in temperature. This means that a large amount of heat energy is required to increase the temperature of a reservoir of water, even if the change in temperature is relatively small. For example, if the specific heat capacity of water were lower, less heat energy would be required to increase its temperature, and the reservoir would heat up more quickly.
What are the factors that affect the heat required to increase the temperature of a reservoir?
There are several factors that affect the amount of heat energy required to increase the temperature of a reservoir, including the mass of the water, the specific heat capacity of the water, and the change in temperature. The mass of the water is a critical factor, as a larger mass of water requires more heat energy to change its temperature. The specific heat capacity of the water is also important, as it determines how much heat energy is required to change the temperature of a given mass of water. Additionally, the change in temperature is a key factor, as a larger change in temperature requires more heat energy. Other factors, such as the thermal conductivity of the reservoir and the surrounding environment, can also affect the amount of heat energy required to increase the temperature of the reservoir.
How can the heat required to increase the temperature of a reservoir be calculated using a calculator?
The amount of heat energy required to increase the temperature of a reservoir can be calculated using a calculator by plugging in the values for the mass of the water, the specific heat capacity of the water, and the change in temperature into the formula Q = mcΔT. The calculator can then be used to perform the calculation and determine the amount of heat energy required. For example, if the mass of the water is 1000 kilograms, the specific heat capacity of the water is 4.184 Joules per gram per degree Celsius, and the change in temperature is 10 degrees Celsius, the calculator can be used to calculate the amount of heat energy required as Q = 1000 kg x 4.184 J/g°C x 10°C = 41,840,000 Joules. This calculation can be performed quickly and easily using a calculator, and can provide a accurate estimate of the amount of heat energy required to increase the temperature of a reservoir.
Deja una respuesta

Entradas Relacionadas